Electrostimulator NEURODENS-PCM (DENAS-PCM 7) of 2020 release. Discontinued! Limited quantity!
$248.00
& FREE Shipping
Quantity
1
Questions? We're here to help
Payment Methods:
About this item
ABOUT PRODUCTS
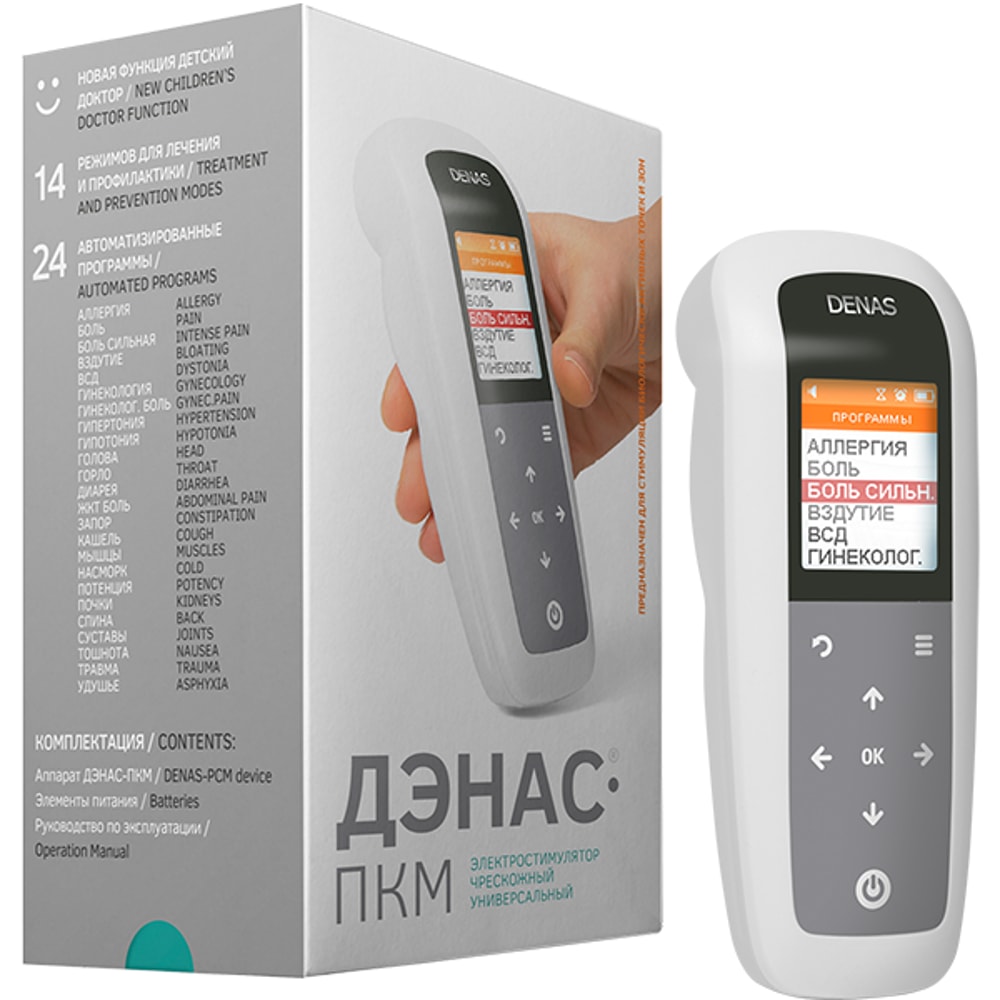
Multipurpose device with a wide range of treatment programs. The device can be used long-term without habituation when having pain, motor disturbances, during rehabilitation after injuries or surgeries.
The device does not require a prescription or compulsory special skills and can be used at home.
The device is safe in service as it uses low voltage current source isolated from the applied part.
Affected areas visualization in the "Programs" menu simplifies learning and usage of the device. Five languages available: English, German, Czech, Italian, Russian.
The kit includes:
• DENAS/NEURODENS-PCM device
• English Manual in pdf format
• English Book "NEURODENS Illustrated Guide to the application" in pdf format
• Consumer packaging
• LR6/AA batteries 2 pcs. not included
Specifications:
• Power supply: 1.5V LR6/AA batteries (2 PCs.)
• Weight: less than 0.35 kg
• Dimensions: 155×50×40 mm
Possibility to set frequencies:
• in "Test" and "Screening" modes – 10 Hz;
• in "Therapy" mode – from1 up to 9.9 Hz – in increments of 0.1; 20, 60, 77, 1077, 2077, 77AM, 125, 140, 200 Hz;
• in "MED" mode – 10 Hz.
Possibility of therapeutic external electrodes connecting.
Warranty period 12 months from the date of sale.
HOW TO USE
When applying the device, take into account that the most pronounced therapeutic effect occurs as a result of course of treatment procedures. For some diseases a course of treatment includes 6-8 procedures, for others it takes 8-12 procedures, and more rarely 14-20 procedures.
There are contraindications. For proper procedures with DENAS devices, as well as for determination of optimal combinations with other treatment methods, read the Operating Manual or consult a specialist.
The following contraindications exist to using a DENAS devices by persons without medical education:
the presence of an artificial heart pacemaker;
the first month of pregnancy;
self-aid in cases of alcoholic intoxication;
individual intolerance.
When the devices is being used by persons with special medical training, all the contraindications are subject to assessment by that person, except where there is the need for urgent operative intervention.
Due to renewal of the registration certificate, the device has not been sold in Russia for a long time. Now it went on sale under a new brand name - NEURODENS. The device itself has not changed (frequency name exception: 7720=2077, 7710=1077).
Note! There is no French language in the new version of the device, but Czech has appeared.
After receiving a new registration certificate, the NEURODENS trademark is used on the packaging, operation manual (RU) and the back of the device, and the previous trademark, DENAS, remains on the device itself. FABERLIC JSC has the exclusive right to sell NEURODENS-PCM.